Beverly Lynds, Skymath Liaison, Unidata Program Center, P.O. Box 3000, Boulder, CO.
http://unidata.ucar.edu/staff/blynds/tmp.html
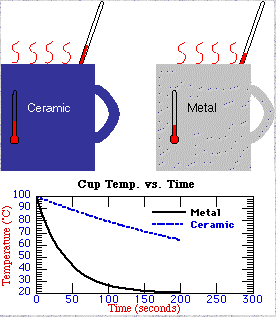
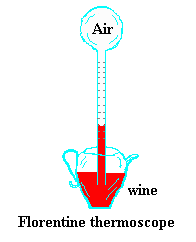
This temperature tutorialx was written for Project
Skymath. The tutorial contains a historical development of temperature
and the kinetic theory and includes the temperature of the universe.
There are lots of links to other sites including NASA's Cosmic
background Explorer Satellite.